Abstract
Introduction The European LeukemiaNet (ELN) 2022 Recommendations adopted the International Consensus Classification (ICC) of Myeloid Neoplasms and Acute Leukemia, which has recently revised the 2016 World Health Organization (WHO) classification of acute myeloid leukemia (AML). In the recent ICC classification, two new categories were added: AML with myelodysplasia (MDS)-related cytogenetic abnormalities category and AML with MDS-related gene mutations category, defined by mutations in ASXL1, BCOR, EZH2, RUNX1, SF3B1, SRSF2, STAG2, U2AF1, or ZRSR2. The 2016 WHO AML with myelodysplasia-related changes (AML-MRC) subtype was no longer an AML category, given that dysplastic morphology, currently used as a criterion for AML-MRC, lacked independent prognostic significance. In this context, we aimed to explore the prognostic significance of AML-MRC subtype, adjusted for mutations in the MDS-related gene group (MDS-RG) and cytogenetic abnormalities. Moreover, we aimed to explore the prognostic significance of mutations in each of MDS-RG group individually.
Methods We conducted a retrospective cohort study at the University of Maryland Greenebaum Comprehensive Cancer Center. All consecutive AML patients treated between 2013-2021 were included. OS was defined as the time from diagnosis to death and was compared between groups using the multivariable Cox proportional hazards (CPH) model and propensity score-adjusted analysis. Moreover, the OS was compared for AML patients categorized by mutational status for each gene of the MDS-RG group, using unadjusted and adjusted analysis. The following baseline characteristics were used to adjust outcomes: age, gender, ethnicity, comorbidities, performance status, 2016 WHO AML type, 2022 ELN cytogenetic abnormalities risk groups, genetic mutations, and treatments administered.
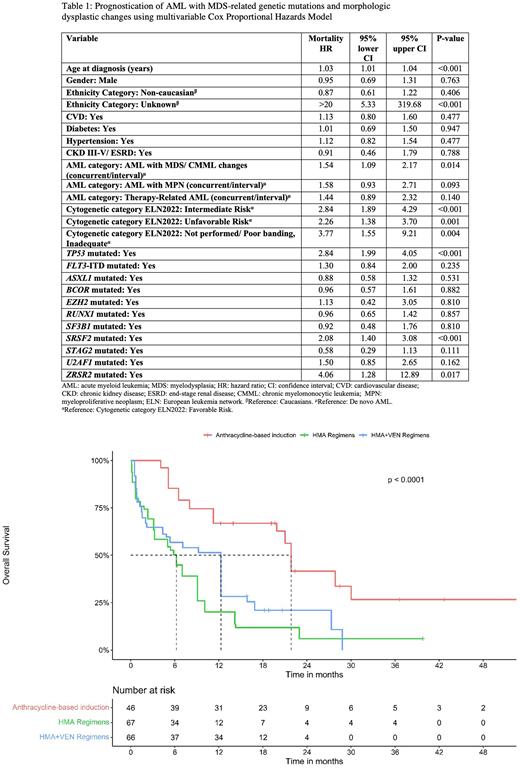
Results Out of 488 AML patients, 222 had myeloid mutation panels at diagnosis and were included in the study. The median age was 64.7 years [IQR 55.7-74.3] and 57% were males. Median follow-up for the whole population was 34.5 months and median OS was 11.9 months (95% CI 9.7-16.8). Frequency of MDS-RG mutations in the whole cohort included: RUNX1: N=42; SRSF2: N=39; ASXL1: N=37; BCOR: N=20; U2AF1: N=15; SF3B1: N=13; STAG2: N=12; EZH2: N=7; and ZRSR2: N=3 patients. When categorizing AML patients by the mutational status of each of the MDS-RGs individually (e.g., ASXL1 mutated AML vs. ASXL1 wild-type AML), patients with mutated genotype were older (~5-10 years), less frequently had de novo AML, and were less frequently treated with intensive chemotherapy. AML patients with mutations in any of MDS-RG group were diagnosed with the 2016 WHO AML-MRC subtype at frequencies ranging from 30 to 60% (e.g., 43% of ASXL1 mutated AML had AML-MRC vs. 20% of ASXL1 wild-type AML had AML-MCR). In the multivariable CPH model (Table 1), AML-MRC was an independent predictor of worse OS (HR: 1.5, 95% CI: 1.1-2.2, P=0.01) after adjustment to covariates, including 2022 ELN cytogenetic abnormality groups and mutations in the MDS-RG group. Only mutated TP53, SRSF2, and ZRSR2 were associated with worse OS (P<0.05). On unadjusted analysis, all patients with mutations in MDS-RGs had a qualitatively lower median OS than patients with wild-type genes, except EZH2, SF3B1and STAG2. Using PS modeling, SRSF2 and EZH2 mutations lower median OS but were not statistically significant. SF3B1 mutation was associated with improved OS (P<0.05). Figure 1 demonstrates adjusted survival curves of AML patients with mutations in any of MDS-RG group treated with anthracycline-based induction or hypomethylating agents +/- venetoclax.
Conclusion The 2016 WHO AML-MRC subclass remained an independent predictor of worse OS, after adjustment for cytogenetic abnormalities and mutations in the MDS-RG group. We found that mutated TP53 predicted worse OS, and mutated SF3B1 predicted improved OS. Further multi-institutional studies are needed to examine the prognostic significance of AML-MRC and mutations in the MDS-RG group.
Disclosures
Baer:Kura Oncology: Research Funding; AbbVie: Research Funding; Forma: Research Funding; Takeda: Research Funding; Kite, a Gilead Company: Research Funding; Ascentage: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal